Pharma 4.0 & Quality: From Apothecary Roots to Big Data Intelligence
Explore how the transition from the "boticário" to Big Data is redefining pharmaceutical quality. Understand the shift to Pharma 4.0 and why qualification is the new strategic frontier.
PHARMACEUTICAL EQUIPMENT
Ariéu Azevedo Moraes
1/20/20264 min read
From Boticários to Big Data: 5 Impactful Truths About Modern Pharmaceutical Quality
In the pharmaceutical industry, precision is the boundary between a life-saving therapy and a catastrophic hazard. A single dosage form represents the culmination of a vast, complex synthesis of chemistry, engineering, and regulatory trust. Historically, the integrity of this process rested in the steady hands of the "boticário" (apothecary), whose artisan skills were the sole safeguard of quality.
Today, we have entered the "Pharma 4.0" era—a "phygital" reality where ancient principles of safety meet high-capacity digital intelligence. As a strategist, I see many organizations struggling to bridge this gap. Understanding industrial quality is no longer just about compliance; it is about mitigating existential commercial and regulatory risks.
1. Calibration Is Not Qualification (And Why the Difference Matters)
In the quest for a state of control, "calibration" and "qualification" are frequently confused, yet relying on calibration alone is a common strategic error that leads to Warning Letters and costly audit findings.
Calibration is a comparative metrological process. It identifies deviations and uncertainties against a known standard. It asks: "Is this equipment measuring correctly (within tolerance) in relation to a traceable standard?" This ensures Metrological Traceability—an essential component of RBC/ISO 17025 accredited standards—but it is only half of the story.
Qualification is a broader risk-management exercise. It asks: "Is this equipment, installed and operated this way, suitable and reliable for this specific process?"
An instrument can be perfectly calibrated but entirely "unqualified" for its environment. For example, a high-precision industrial scale may be calibrated to international standards, but if it is placed in a drafty area subject to air currents or used outside its tested range, the data it generates is a regulatory liability. In a "Smart Factory," an unqualified instrument is a broken link in the digital chain.
"Is the equipment measuring correctly...?" vs. "Is this equipment... suitable and reliable for this specific process?"
--------------------------------------------------------------------------------
2. Pharma 4.0: Moving Toward a "Phygital" Reality
We have moved beyond the Third Industrial Revolution’s focus on basic Information Technology into the Fourth: Pharma 4.0. This is the era of the "Smart Factory," where systems are integrated, autonomous, and self-managed. The core technologies driving this shift include:
Internet of Things (IoT): The backbone of connectivity, allowing machines to communicate in real-time.
Artificial Intelligence (AI): Algorithms that handle autonomous decision-making and fraud detection.
Cloud Computing: Ensuring data is accessible to all stakeholders instantly, regardless of geography.
Cyber-Physical Systems (CPS): The integration of computational logic with physical production steps.
Big Data and Analytics (BDA): Far more than a marketing tool, BDA is vital for monitoring product behavior and making real-time process adjustments to ensure consistency.
This transition enables "mass customization" and real-time data integration with regulatory bodies, moving the industry away from rigid, reactive production models.
--------------------------------------------------------------------------------
3. The URS is the Project’s Birth Certificate
The User Requirements Specification (URS, or ERU) is the most critical risk-mitigation tool in a strategist's arsenal. It serves as a strategic procurement safeguard against "technical debt" and supplier-imposed solutions.
Whether you are acquiring a new reactor or a complex computerized system, the URS bridges the gap between the user’s operational needs and technical engineering solutions. It must be a multidisciplinary document involving production, quality, and engineering. Crucially, a URS is only valid when developed after a thorough risk analysis, ensuring every critical quality point is addressed before a single dollar is spent.
ANVISA defines the URS as: "A set of owner, user, and engineering requirements necessary and sufficient for the creation of a feasible project with the intended purpose for the system."
--------------------------------------------------------------------------------
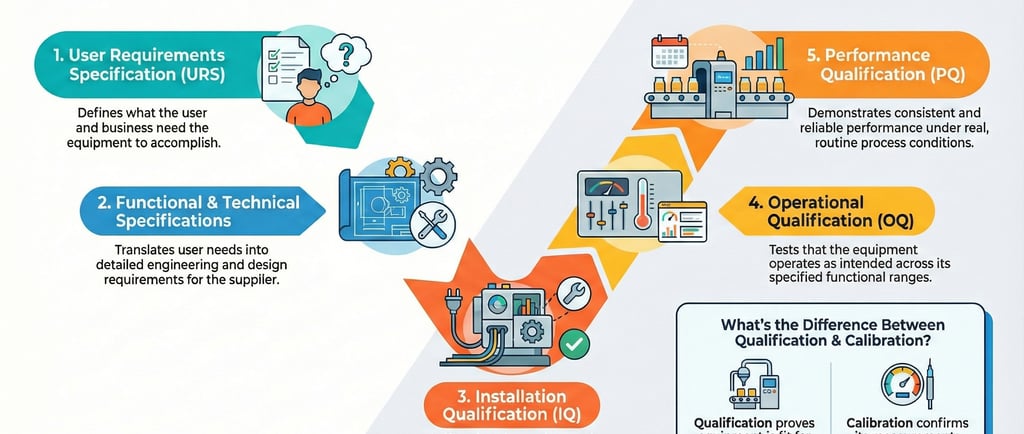
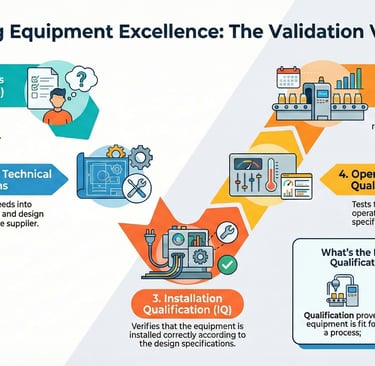
4. The "V-Model" and the Rigor of Validation
Validation is not a one-off event; it is a documented lifecycle. At the macro-level, this is governed by the Validation Master Plan (PMV), the foundational document that defines the hierarchy and strategy of all studies.
The industry standard for execution is the "V-Model," which provides a structural map connecting requirements to testing. A critical nuance of the V-Model is that the initial URS serves as the direct benchmark against which final Performance Qualification (PQ) is measured.
The three pillars of this rigor are:
Installation Qualification (IQ): Verifying the environment and infrastructure (power, air, placement) meet manufacturer standards.
Operational Qualification (OQ): Testing controls, software interlocks, and alarms across the full operating range.
Performance Qualification (PQ): Proving consistent results under real-use conditions. A standard example is thermal mapping in drying ovens, ensuring uniform heat distribution when the unit is fully loaded with product.
By adhering to this model, companies mitigate risks like cross-contamination and product loss, protecting the brand's reputation and the consumer’s health.
--------------------------------------------------------------------------------
5. The Evolving Role of the Pharmacist
The pharmacist’s journey has evolved from the artisan "boticário" to the modern Data Integrity Steward. In 1877, the adaptation of the Brockedon machine for tablet production by John Wyeth & Brother shifted the pharmacist from the bench to the industrial floor.
In Pharma 4.0, the pharmacist's role has been elevated. As machines take over the "How"—the execution and autonomous maintenance through AI—the pharmacist must manage the "Why." This requires "thoughtful synthesis" and "critical thinking." The modern industrial pharmacist is no longer a manual operator but a strategist who ensures that these complex, autonomous digital systems remain in a state of control and compliance.
--------------------------------------------------------------------------------
Conclusion: The Future of Quality is Integrated
Quality is not achieved by calling a technician once a year for a certificate. It is a continuous, documented lifecycle that integrates technology, human expertise, and regulatory strategy.
As we move deeper into the Pharma 4.0 era, every leader must ask: Are your current processes truly "qualified" for the digital age, or are they simply "calibrated" for a world that no longer exists?
A structured program of calibration and qualification is not a cost center; it is an investment in market longevity. The cost of a structured program is exponentially lower than the price of a single discarded batch, a regulatory fine, or a compromised brand.
Contact
Reach out for domain inquiries today
Phone
© 2025. All rights reserved.